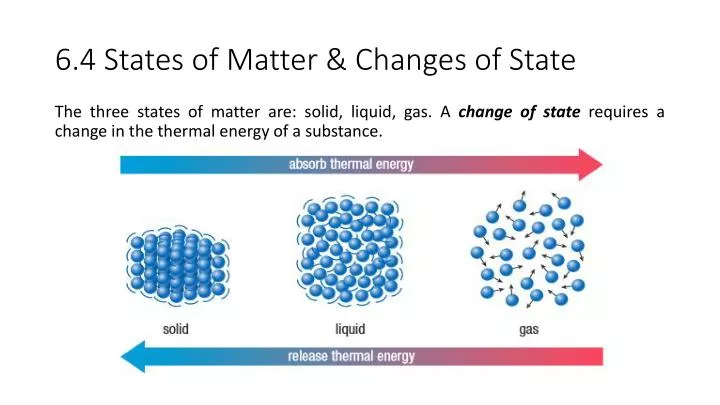
There is a positive correlation between temperature and the movement of molecules. Similarly, when the temperature is increased the movement of molecules in the substance will increase because increases in temperature with heat breaks the bonds and physical forces that hold the molecules tightly together are broken conversely, when the temperature is decreased the movement of molecules in the substance will decrease because decreases in temperature with the application of cold will hold the molecules more tightly together. In other words, the greater the pressure, the less movement of molecules and the lesser the pressure, the more movement of molecules in matter.Īs pressure increases, molecular movement decreases.Īs pressure decreases, movement increases. There is a negative correlation between pressure and the movement of molecules. When the pressure is increased the movement of molecules in the substance will decrease because increases in terms of pressure hold the molecules in matter more tightly together conversely, when the pressure is decreased, the movement of molecules in the substance will increase because decreases in terms of pressure allows the molecules in matter further away from each other. The movement of molecules in matter changes and varies according to the variation in terms of temperatures and pressure. The most dramatic, most rapid random colliding movements of molecules occur in gases. This movement is simply small vibrations in solids because the molecules in solids are more tightly packed together than they are in liquids and gases. The word kinetic is derived from and defined as movement. The Kinetic Molecular Theory states that molecules in the states of matter move. 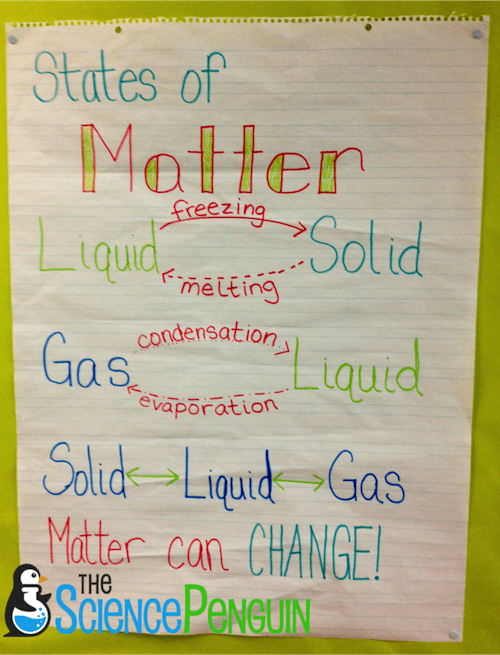
The Changing States of Matter Kinetic Molecular TheoryĪs previously discussed somewhat in the previous section, atoms and molecules make up matter. Desublimation: The alternative term for deposition.Deposition: The transition of a gas directly to the solid state without passing through, or transition to, the liquid state.Sublimation: The transition of a solid to a gas without going through the intermediate transition step of transforming into a liquid.Melting: The transition of a solid into a liquid.Freezing: The transition of a liquid to a solid. 
Condensation: The transition from a vapor or gas back to a liquid. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
